Mid-cycle, the phone started vibrating.
The April 15 HHS action — Secretary Robert F. Kennedy Jr. directing the FDA to remove twelve compounds from Category 2 of the 503A bulk drug substances list, the federal designation that had barred compounding pharmacies from legally handling them since 2023 — made a compound I had pushed into my bloodstream at six that morning, and half a dozen others I had been running for years, into something legally different overnight. Not different chemically. The molecules didn’t change. The paperwork did. Peptide legalization 2026 is now a sentence you can say out loud in a clinic without a lawyer flinching.
This matters less than the coverage will suggest. It also matters more.
What the Action Actually Did
Before April 15, peptides occupied one of the stranger legal positions in American pharmacology. Most of the compounds used by serious practitioners — BPC-157, TB-500 fragment, GHK-Cu, Epitalon, MOTS-c, Ibutamoren, Semax — lived in a regulatory purgatory. They were not FDA-approved finished drugs. They were not controlled substances. And since September 2023, they were not compoundable: the Biden-era FDA had placed them on the 503A “Category 2” bulk substances list — formally designated Bulk Drug Substances that Raise Significant Safety Risks — which prohibited licensed compounding pharmacies from sourcing or dispensing them. (Source: FDA 503A Bulk Drug Substances Nominated for Use in Compounding — Category 2 list, as of 2023)
The practical effect was a contradiction. A physician could, in principle, write a prescription for BPC-157 to a patient with a documented injury. But because the FDA had placed it in Category 2, no 503A pharmacy would fill it. The compound still existed. The prescribing authority still existed. The legal pathway to get from one to the other did not. This is how a pharmacological community ends up buying its medicine from vendors whose product pages carry an explicit not for human use disclaimer next to vials that are clearly destined for humans.
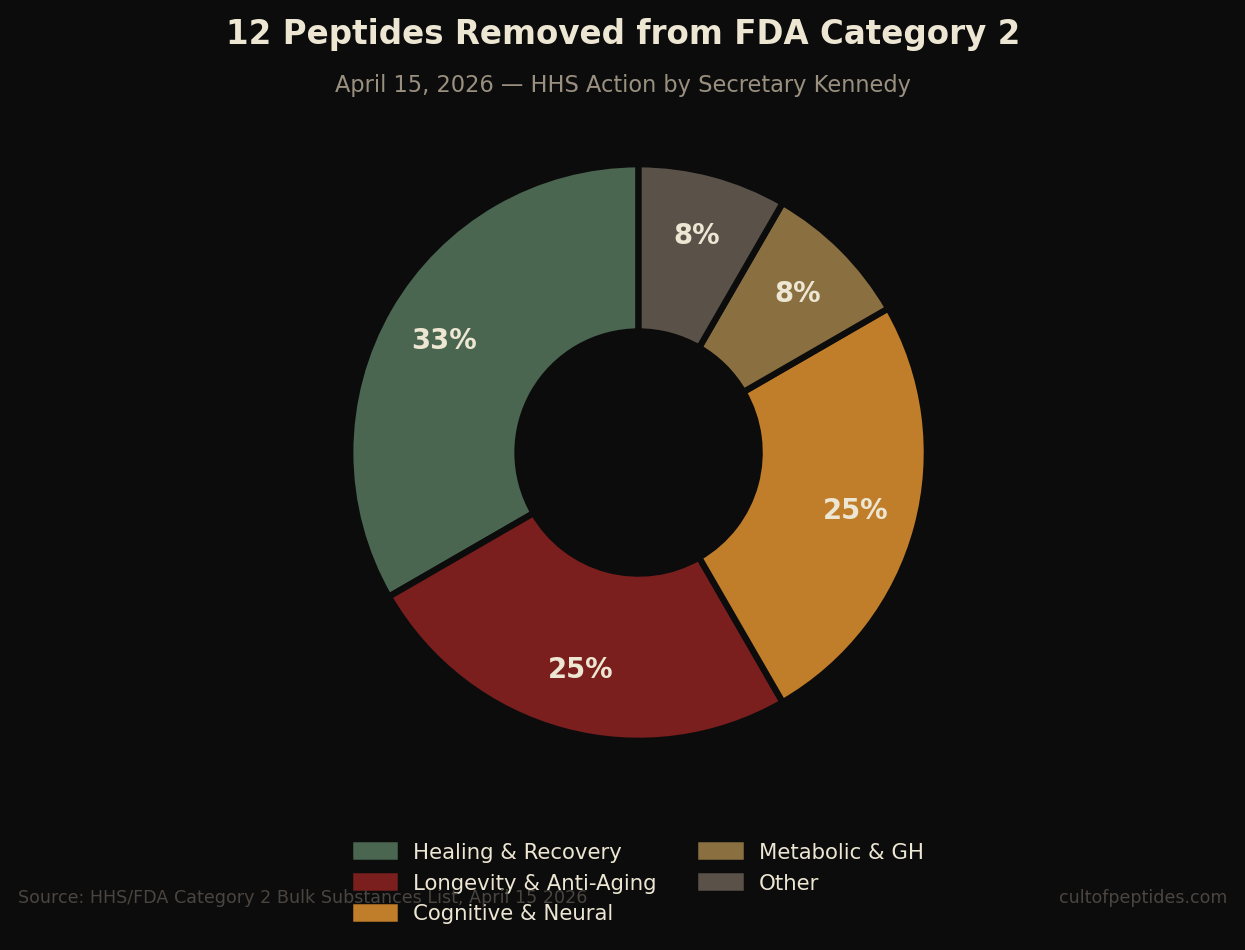
The April 15 action reversed this through an administrative directive. After nominators withdrew all twelve peptides from the Category 2 designation, Secretary Kennedy directed the FDA to remove them from the restricted list. The twelve compounds now cleared from Category 2 are:
- BPC-157
- Thymosin beta-4 fragment — LKKTETQ (the active fragment of TB-500)
- Epitalon
- GHK-Cu (injectable)
- MOTS-c
- DSIP (Emideltide)
- Dihexa Acetate
- Ibutamoren Mesylate (MK-677)
- Melanotan II
- KPV
- Semax (heptapeptide)
- Cathelicidin LL-37
Under the new framework, each compound is scheduled for review by the Pharmacy Compounding Advisory Committee (PCAC) beginning at its next two meetings, starting in July 2026 — where, per Kennedy’s statement, “independent experts will rigorously evaluate each substance on its scientific merits using full clinical, pharmacological, and safety evidence.”
What this means in practice: the prohibition has been lifted. The compounds are no longer barred from compounding pharmacies on safety grounds. They are not yet on the Category 1 positive list that gives full explicit clearance. They exist, for now, in a transitional state that is meaningfully more permissive than the past two years — and will be resolved by PCAC evaluation later this year.
What this is not: FDA drug approval. What the action grants is removal of a specific prohibition on compounding — not a finding of efficacy. No one has proved BPC-157 heals human tendons to the FDA’s approval standard. That trial has not been run. (Source: ClinicalTrials.gov — current registered human trials for BPC-157, April 2026) What has changed is that a physician can now prescribe it and a compounding pharmacy can fill that prescription without legal exposure. Whether they should, for any given patient, is a medical question the action does not answer.
The distinction between legal to compound and proven to work is the distinction most of the coverage is about to collapse. Hold on to it. Everything downstream depends on keeping those two ideas separate.
What Actually Changes for Practitioners
For the cohort of readers this publication was built for — people who were running protocols before Thursday — the changes are narrower than the headlines suggest, and more meaningful than they first appear.
Access through regulated pharmacies is now possible — with a caveat. Until this week, a serious person running a BPC-157 cycle for a documented tendon injury had to choose between a research peptide from a vendor with variable quality control, or nothing. Starting now, a clinician familiar with these compounds can write a prescription, and a compounding pharmacy can fill it. The caveat: the PCAC evaluation process runs through the latter half of 2026. Most well-run compounding pharmacies will move cautiously until the Category 1 positive listing is formally confirmed. The first regulated scripts will be written this month. The system will not be fully fluid until the PCAC process completes.
Not every compound in a typical practitioner’s stack is on this list. This is the detail most coverage will miss. Ipamorelin, CJC-1295, IGF-1 LR3, and Tesamorelin — compounds widely used in serious stacks — are not on the twelve that were removed from Category 2. Tesamorelin is separately FDA-approved as Egrifta. The others exist in a different part of the regulatory landscape and are unaffected by this action. Before assuming your entire protocol is now pharmacy-accessible, read the specific list.
Cost will rise before it falls. A compounded peptide from a regulated pharmacy, prescribed by a physician with liability insurance, moving through a benefit manager, will not compete on price with a $35 vial shipped from a research vendor. First movers will price for quality signaling. Expect six to twelve months of premium pricing before bulk supply chains stabilize and competition normalizes rates. For practitioners who have been buying through informal channels for years, the regulated pathway will initially feel like paying four times as much for the same molecule. That is accurate. It is also what a regulatory system looks like.
The physician bottleneck doesn’t disappear — it moves. Most primary care doctors have never heard of BPC-157 and are not going to prescribe it based on a patient’s printed PubMed results. The bottleneck is no longer legal; it is now educational. Expect a fast expansion of specialty clinics in the anti-aging, longevity, and sports medicine spaces that already know these compounds. Expect the major telehealth platforms — Hims, Ro, Function Health-adjacent services — to launch peptide lines within ninety days. Some will be excellent. Most will not. The differentiator will be whether the clinician can actually name the rat studies the dosing is extrapolated from.
Documentation shifts from optional to essential. A peptide protocol that was previously unverifiable is now, in theory, legible to the medical system. Bloodwork, baseline metrics, symptom journaling, contraindication screening — these were always the right approach. They are now the only defensible approach. What was personal becomes medical, and the medical system has strong opinions about documentation. Anyone serious about this space should have been keeping records already. Those who weren’t now have a reason to start.
What’s Just Noise
The next six weeks will produce a certain kind of content. Affiliate-driven “best peptides” rankings. Influencers claiming the compound that reset their skin has been finally exonerated. Supplement brands repackaging legal peptides as proprietary blends with “clinically studied” stamps on the label. A great deal of it will look like journalism. Almost none of it will be.
Here is what the April 15 action did not do.
It did not make peptides safe for everyone. The evidence base for BPC-157 — to take the most cited example — remains overwhelmingly rodent-based. (Source: Sikiric et al., “Brain-gut Axis and Pentadecapeptide BPC 157,” Journal of Physiology Paris, 2012, and subsequent animal studies) Human clinical data for most compounds on the new list is limited, surrogate-endpoint, or drawn from small trials. A regulatory status change is not a risk profile. Anyone with active malignancy, significant cardiac disease, an endocrine disorder, or who is pregnant should be having a different conversation with a different kind of doctor. The compounds most strongly associated with off-label longevity use — growth hormone secretagogues in particular — have interaction profiles with existing malignancies that should not be brushed past.
It did not settle the science. The molecules do things. What exactly they do in human bodies at the doses most readers of this publication are running is, for several of them, meaningfully underspecified. Community protocols are extrapolations from animal data or from clinical trials using different compounds at different doses. This is not, in most cases, dangerous. It is also not the same thing as knowing what you are doing. Anyone claiming otherwise is selling something.
It did not solve the vendor quality problem. The difference between a peptide synthesized at 99% purity with verified mass spectrometry and one at 85% with unknown endotoxin load is the difference between a useful protocol and a potentially harmful one. Bulk supply flowing into 503A pharmacies will be subject to USP standards, but the transition will be chaotic, and a physician’s prescription pad is not a quality guarantee. The question that now matters: which compounding pharmacy is filling this? Ask for the certificate of analysis. Ask about the API supplier. The serious pharmacies will answer without flinching. The others will get evasive.
It did not resolve the dosage question. Clinical literature for peptides is sparse, and most of it uses doses calibrated to species that weigh less than a housecat. The community protocols circulating in forums and group chats are extrapolations, not guidelines. They are not, in general, wrong — many practitioners have been running them safely for years. But they are also not what any serious trial has studied. Anyone reporting on peptide dosage should be clear about this distinction. Most will not.
It did not make the RFK peptides legal action a closed case. The PCAC evaluation begins in July. The outcome of those committee meetings — whether each compound gets cleared, restricted, or returned to a constrained category — is not predetermined. Kennedy’s statement frames these as reviews “on scientific merits using full clinical, pharmacological, and safety evidence.” Some compounds will fare better than others. Melanotan II, for instance, has a different safety profile than BPC-157, and the committee is unlikely to treat them identically. The list of twelve is not a list of twelve approvals. It is a list of twelve compounds under active regulatory review.
Why This Moment Actually Matters

The legalization of peptides does not matter because peptides are suddenly legitimate. They were legitimate before. In the research literature. In competitive athletics. In the private practices of physicians who quietly understood what their wealthier patients were running and treated those patients with more sophistication than they would admit to their licensing boards. The conversation existed. What changed on April 15 is that the conversation left the basement.
Which means the conversation is now up for grabs.
I saw it at work. Scrolling between calls, RFK’s post came across the feed, and my first thought was not vindication. It was that the first step had finally happened, and first steps are the hard ones. I screenshotted it into the same group chat I had been boring about peptide companies for years.
The next five years will determine who gets believed about peptides. The serious voices — the physicians who have been prescribing responsibly, the researchers who have run the studies, the practitioners who have paid close attention — are not, historically, the loudest. The loudest have typically been the supplement industry, influencer-adjacent clinics, and content farms optimizing for affiliate revenue rather than clinical endpoints. That calculus does not change unless someone decides to do the work.
This is why Cult of Peptides exists.
We are not a supplement company. We will not have a proprietary blend. We will not publish “seven peptides every man needs after forty.” We will cover this beat the way it deserves to be covered — slowly, carefully, cited, and in public. When we run a protocol, we will tell you the compound, the dose, the duration, the cycle structure, and the outcomes — measured, not remembered. When the evidence contradicts community wisdom, we will say so. When community wisdom contradicts the evidence, we will say so. When we do not know something, I don’t know will appear in print, because that is a phrase that builds trust.
The publication’s tenets were not designed to be pretty. They were designed to be operating constraints. Protocol before product. Cite the study or skip the claim. First-person or no-person. Readers, not customers. Vague is violence.
May support. Could help. Some studies suggest. These phrases are ubiquitous in health content because they are escape hatches — the language that lets a writer sound authoritative without saying anything a lawyer would ever have to defend. Vague is the architecture of grift. Specific is the architecture of trust. Peptide legalization 2026 is not the story. It is the beginning of the story. The story is what happens next — who shows up to cover it with rigor, who shows up with a supplement affiliate link, and which voices the serious practitioner chooses to trust when both are available.
What Practitioners Are Actually Asking
Are peptides now legal in the US?
Specific peptides — the twelve removed from FDA Category 2 by the April 15, 2026 HHS action — are no longer barred from compounding pharmacies on safety grounds. They remain prescription medications, not over-the-counter supplements, and each is scheduled for formal PCAC evaluation beginning July 2026. The RFK peptides legal action does not make these compounds available without a prescription. It removes the specific prohibition that prevented licensed compounding pharmacies from filling prescriptions for them.
Which peptides does the 2026 action apply to?
The twelve compounds removed from Category 2 are: BPC-157, Thymosin beta-4 fragment (LKKTETQ), Epitalon, GHK-Cu (injectable), MOTS-c, DSIP (Emideltide), Dihexa Acetate, Ibutamoren Mesylate (MK-677), Melanotan II, KPV, Semax (heptapeptide), and Cathelicidin LL-37. Compounds not on this list — including Ipamorelin, CJC-1295, and IGF-1 LR3 — remain in their prior regulatory status. Coverage conflating “peptides are legal” with “all peptides are legal” is not accurate. The list is the single most important document for anyone trying to understand what actually changed.
Do you need a prescription for peptides under the new framework?
Yes. The 2026 action did not make peptides available over the counter. It removed the compounding prohibition for twelve specific compounds. A physician still needs to prescribe. A pharmacy still needs to compound and dispense. In practice, specialty clinics — anti-aging, integrative medicine, sports medicine, longevity-focused practices — are likely to be the first scalable access points for most patients in the near term.
What does this mean for compounding pharmacies?
Considerable new business after an adjustment period. 503A pharmacies (traditional compounders serving individual patient prescriptions) and 503B outsourcing facilities are the primary legal fulfillment pathway under the new framework. Most cautious compounders will wait for PCAC clearance before actively marketing these compounds, but the legal barrier to filling existing prescriptions has been removed. When the system stabilizes, ask which pharmacy your prescription is going to — and ask for the certificate of analysis — before the prescription is written.
This story is not a news cycle. It is an inflection. Peptides have been around in one form or another since insulin. Serious human interest in them as optimization tools is nearly a quarter-century old. The April 2026 moment is the point at which the regulatory system caught up with what thousands of practitioners were already doing quietly — and that means a great deal is about to happen, in public, fast. We are going to cover all of it.
If you want to follow the story properly, join the Congregation. Weekly dispatches. Protocol updates. Nothing else.