What Twelve Weeks of BPC-157 Actually Did to My Shoulder
The peptide that launched a thousand protocol threads is also one of the most misrepresented molecules in this space. Let me be precise about both of those things.
BPC-157 — Body Protection Compound 157 — is a synthetic pentadecapeptide derived from a protein found in human gastric juice. Fifteen amino acids. Stable in acidic environments. Not endogenous in the form most people are injecting or swallowing. The “derived from” framing that circulates in forums collapses an important distinction: presence of a parent protein in gastric secretions is not the same as the body producing this specific sequence at therapeutic concentrations. Keep that distinction close.
This is article two in this publication’s founding sequence. The first piece covered the legal architecture around compounding. This one covers the molecule most practitioners in this space run first, run longest, and understand least precisely.
What the Rodent Literature Actually Shows
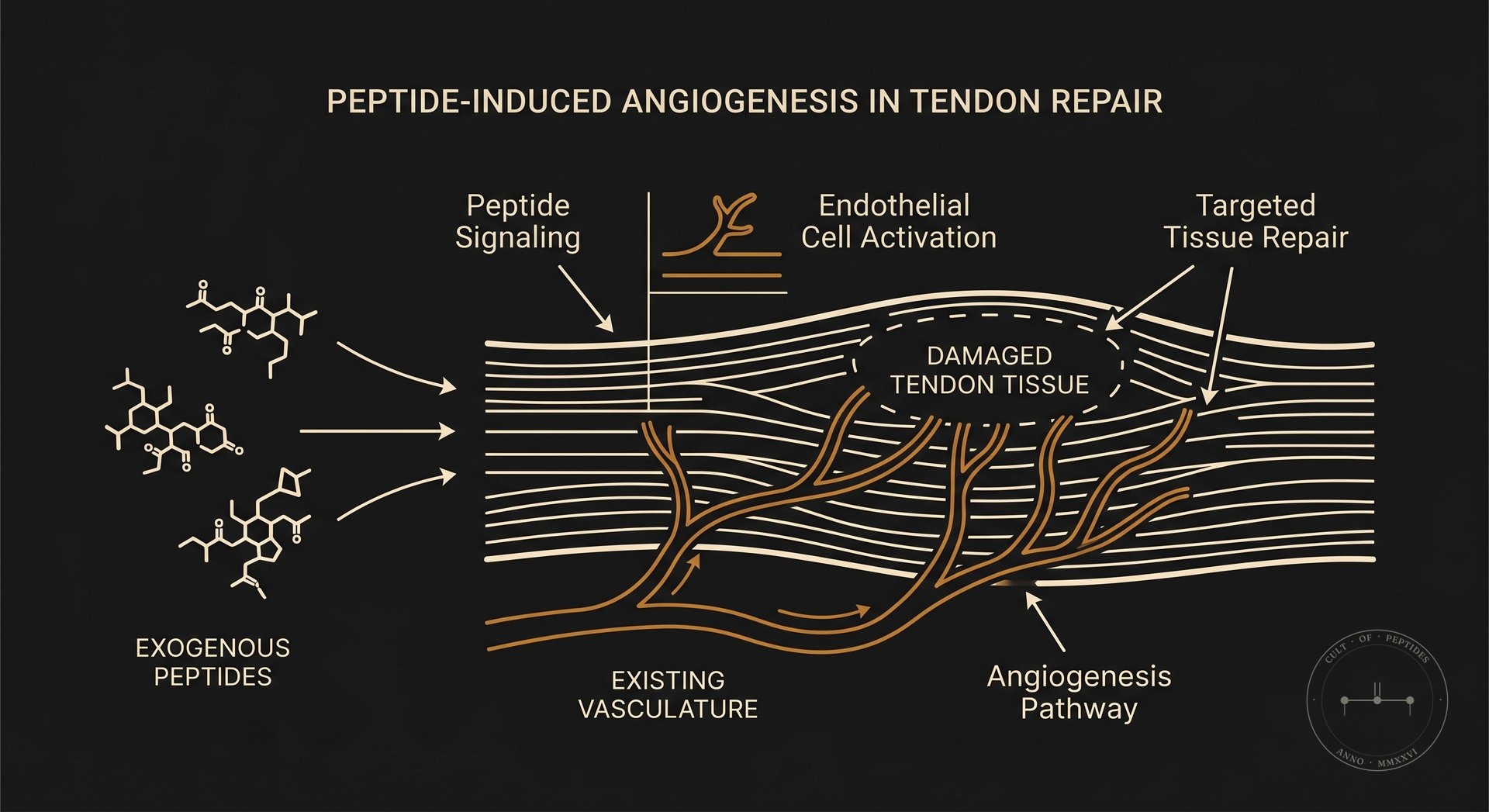
The honest account of BPC-157’s evidence base starts with a single uncomfortable fact: almost everything we know comes from rat models, and almost all of that research originates from one laboratory — the group led by Predrag Sikirić at the University of Zagreb.
That is not disqualifying. Sikirić et al. have published prolifically and consistently across decades. A 2014 study in Molecules (Chang et al.) documented upregulation of growth hormone receptor expression in rat tendon fibroblasts treated with BPC-157 — increases up to sevenfold at day three — alongside enhanced cell proliferation when growth hormone was added to pretreated cells. A 2016 review in Current Neuropharmacology (Sikirić et al.) documented that BPC-157 counteracts gastrointestinal and liver damage from NSAIDs including ibuprofen, diclofenac, and paracetamol, consistent with its proposed cytoprotective mechanism.
The mechanistic picture that emerges from this body of work is internally consistent: BPC-157 appears to modulate nitric oxide signaling, promote angiogenesis at injury sites, and interact with the growth hormone/IGF-1 axis in ways that accelerate tissue repair. The same 2016 Current Neuropharmacology review documented BPC-157’s interaction with both NOS substrate (L-arginine) and NOS blocker (L-NAME), with protective effects across multiple rat models partially dependent on this pathway.
What this literature does not provide: human RCT data on musculoskeletal healing outcomes. What it does not tell us: optimal human dosing, bioavailability differences between subcutaneous injection and oral administration in humans, or long-term safety signal data across any population.
The rodent evidence is suggestive and mechanistically coherent. It is not a protocol. Do not use it as one.
The Oral vs. Injectable Question Is Not Resolved
This is the question I see collapsed most often in forum discussions, and the collapse matters.
The argument for oral BPC-157 rests on its gastric stability — unlike most peptides, BPC-157 does not appear to degrade rapidly in an acidic environment. Rat studies support systemic effects from oral administration. A 2023 review in Pharmaceuticals (Sikirić et al.) documented BPC-157’s stability in human gastric juice and its effectiveness via oral administration across parenteral, intragastric, and per-oral routes in multiple models. The mechanism proposed: local action combined with some degree of systemic absorption.
The argument against treating oral and injectable as equivalent: we have no pharmacokinetic data in humans establishing bioavailability for either route. In rats, subcutaneous and intraperitoneal administration produce measurable plasma concentrations. Whether oral administration in humans produces systemic concentrations sufficient to affect tendon healing or neurological outcomes — the use cases most practitioners care about — is not established. The gastric stability data tells us the molecule survives the stomach. It does not tell us how much crosses into systemic circulation, at what concentration, or with what half-life.
I am not arguing against oral administration. I am arguing against the inference that gastric stability equals clinical equivalence to injection. Those are different claims.
What the Mechanism Suggests About Use Cases
Working from the mechanistic literature rather than claimed outcomes, here is what the evidence supports as plausible targets:
Gastrointestinal cytoprotection. This is the most evidence-proximate use case. The compound was originally studied in the context of gastric ulcer protection, and the rat literature here is the deepest. If there is a human analogue to the gastric protection seen in rodent models, oral administration is the logical route.
Musculoskeletal injury repair. The tendon and ligament healing data in rodents is consistent across multiple Sikirić group papers and has been partially replicated by independent groups. A 2011 study in Journal of Applied Physiology (Chang et al.) showed improved tendon healing in a rat model involving tendon outgrowth, cell survival, and cell migration — independent replication outside the Zagreb group. The mechanism involves VEGF upregulation and collagen organization at the repair site. Whether this translates to human healing timelines at the doses circulating in the community remains unknown.
Neurological and mood effects. This is where the evidence thins considerably. Rat models have shown anxiolytic effects and dopaminergic modulation. The same 2016 Current Neuropharmacology review (Sikirić et al.) documented anxiolytic properties, antidepressant activity, and interaction with dopaminergic and serotonergic systems in rat models. Human data here is absent. The gap between dopaminergic modulation in a rat brain and clinical mood effects in a human nervous system is not a gap the current literature crosses.
I am not saying these effects don’t occur in humans. I am saying the evidence chain for them is: rat model → plausible mechanism → anecdote. That is a chain. It is not proof.
The Legal Status Question Intersects With the Evidence Gap
BPC-157’s legal situation — as I covered in the first piece — sits in the compounding gray zone. It is not FDA-approved. It is not on the FDA’s bulk substances list for 503A compounding. It is not categorically illegal to possess. The regulatory status reflects, at least in part, the evidence status: a molecule without human trial data sufficient for approval, circulating in a market that has outrun the research.
The practical consequence for practitioners: there is no pharmaceutical-grade reference standard for human dosing. The dosing protocols circulating in the community — typically 200–500 mcg per day subcutaneously, 4–6 week cycles, though these numbers vary widely by source — are derived from rat study dose conversions and community experimentation, not from phase II or phase III trial data. I want to be clear that I am reporting what circulates, not endorsing a protocol I cannot source to a controlled trial.
The compounding pharmacies producing BPC-157 are operating under significant regulatory uncertainty. What that means for purity, concentration accuracy, and stability is a variable the buyer cannot fully control from the outside.
What I Run and What I Don’t Know About It
I have used BPC-157 subcutaneously following a partial ligament injury. I ran it for six weeks at the lower end of the circulating community range. The injury resolved on a timeline that my orthopedist described as faster than typical — a statement that carries exactly zero controlled evidence weight, because I was also doing physical therapy, sleeping eight hours, and eating aggressively toward protein targets during the same period.
I don’t know what the peptide did. I know what the timeline was. Those are different things, and I think collapsing them is how this space manufactures its most confident and least defensible claims.
What I believe, based on the mechanistic literature and the consistency of the rodent data: there is a real biological effect here that warrants serious human investigation. What I think is happening in most practitioner-level anecdotes, including mine: a signal buried in so much noise that individual n=1 reports are nearly uninterpretable.
That is not a reason to dismiss the molecule. It is a reason to hold the claims precisely.
What Practitioners Are Actually Asking
Is BPC-157 safe for human use? The rodent safety profile is clean across a substantial body of research — no significant toxicity signals at therapeutic doses in animal models. Human safety data from controlled trials does not exist. What circulates in the community is accumulated anecdote across years of self-experimentation. The absence of widely reported serious adverse events is weakly reassuring. It is not a safety profile.
Does the oral form work as well as injectable? The rodent literature shows systemic effects from oral administration, but no human pharmacokinetic data establishes bioavailability for either route in people. Oral may work for gastrointestinal applications where local action is part of the mechanism. Whether oral produces systemic concentrations sufficient for musculoskeletal or neurological effects in humans is not established.
What dose should I run? I will not give you a number. There is no human RCT establishing an effective dose. What circulates — typically 200–500 mcg/day subcutaneously — is extrapolated from rodent studies and refined through community experimentation. Anyone presenting this as settled dosing science is overstating the literature.
How long does a cycle need to be? No controlled data on cycle length exists for humans. The community convention of four to six weeks appears to be derived loosely from injury repair timelines in rodent models and refined through self-report. It is a reasonable starting framework. It is not evidence-based in any meaningful clinical sense.
Can BPC-157 help with depression or anxiety? The rodent evidence for dopaminergic modulation and anxiolytic effects is real. The evidence chain from that data to human clinical outcomes is: animal model to mechanism to anecdote. I cannot responsibly call that evidence of efficacy for mood disorders in humans. The question is worth serious investigation. It has not received it.
The molecule is interesting. The literature is suggestive. The human evidence is absent in the places that matter most to the people running it. That gap is not a reason to stop paying attention — it is the reason to pay more careful attention than the forums typically do.
