What Peptides Are, and Why the Answer Is More Interesting Than You’ve Been Told
Most introductions to peptides start with a definition that sounds like it was written for a supplement label. This one won’t. If you’re here because something changed legally in the last year and you want to understand what all the noise is actually about, start here.
A peptide is a chain of amino acids. That’s the definition. It’s also almost useless on its own, because proteins are chains of amino acids too, and so is the collagen in your joints, and so is insulin, and so is the hormone your pituitary gland releases at 2 a.m. to trigger a pulse of growth hormone. The definition is technically correct and practically insufficient. What matters is what peptides do — and more specifically, what a narrow subset of them do when introduced to a human body at therapeutic doses.
That’s what this article is about.
The Biochemistry, Without the Lecture
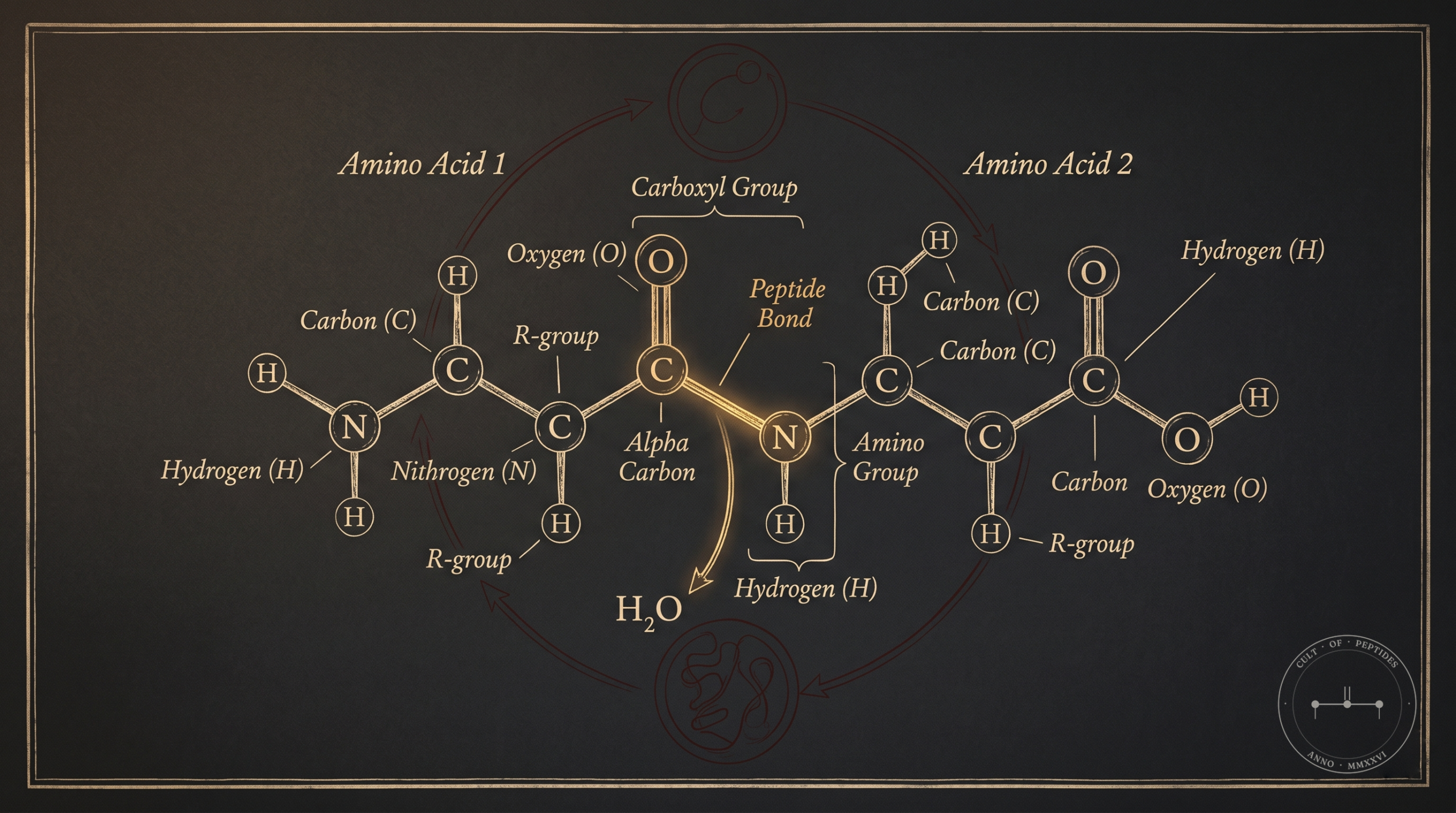
Amino acids are the building blocks of all proteins in the human body. There are 20 standard amino acids. Chain two together and you have a dipeptide. Chain three and you have a tripeptide. Somewhere above roughly 50 amino acids, convention says you’ve crossed into protein territory — though that boundary is fuzzy and somewhat arbitrary.
What makes peptides biochemically interesting isn’t their size. It’s their specificity. A short peptide chain can be engineered — or discovered in nature — that binds to a very precise receptor, triggers a very precise signaling cascade, and produces a very precise physiological effect. Large proteins do many things at once. A targeted peptide can do one thing, loudly.
That specificity is the whole game.
How Peptides Actually Signal
Most therapeutic peptides work through one of two mechanisms: receptor binding or signaling pathway modulation.
Receptor binding is what it sounds like. The peptide has a shape that fits a specific receptor on a cell surface. When it binds, it either activates or blocks whatever that receptor normally does. Sermorelin, for example, binds to the growth hormone-releasing hormone receptor (GHRHR) in the pituitary gland and stimulates growth hormone secretion. It doesn’t introduce exogenous growth hormone — it asks your pituitary to make more of its own. That distinction has clinical relevance, and we’ll come back to it.
Signaling pathway modulation is less direct. Some peptides appear to upregulate or downregulate cellular repair pathways without cleanly fitting the receptor-binding model. BPC-157 — a peptide derived from a protein found in gastric juice — is the most studied example of this. In rat models, it has shown consistent effects on angiogenesis (the formation of new blood vessels), nitric oxide signaling, and growth factor expression (Sikiric et al., Current Neuropharmacology, 2016). The mechanism isn’t fully characterized. That’s not a sales pitch; it’s a limitation that deserves to be named.
Why Size Matters for Delivery
Here’s a practical consequence of the chemistry: peptides are broken down in the gut. Most of them. Your digestive enzymes don’t know the difference between a therapeutic peptide and a piece of chicken breast. Both are amino acid chains. Both get hydrolyzed.
This is why most therapeutic peptides are administered subcutaneously — small injections under the skin, typically into abdominal fat — rather than taken orally. The peptide bypasses the digestive system, enters circulation intact, and reaches its target tissue. Some peptides have been formulated as oral or nasal compounds, but for the majority of compounds discussed in serious clinical or research contexts, subcutaneous injection is the delivery method.
That’s not a minor detail. It’s why this conversation looks different from the one about supplements.
The Difference Between a Peptide and a Supplement
Walk into any sports nutrition store and you’ll find collagen peptides, creatine, amino acid blends. These are all technically peptides or peptide precursors. They’re also nothing like what practitioners in this space mean when they say “peptides.”
The distinction comes down to pharmacokinetics and mechanism. A collagen peptide supplement provides substrate — raw material your body may or may not use. A therapeutic peptide acts as a signal. It doesn’t add building blocks. It changes what the building blocks are told to do.
That distinction matters even more in peptides for skin, where cosmetic peptides, collagen supplements, topical GHK-Cu, and injectable GHK-Cu often get flattened into the same conversation despite having different mechanisms and evidence standards.
Insulin is a peptide. Oxytocin is a peptide. GLP-1 agonists like semaglutide — which reshaped the entire conversation about obesity medicine — are peptide analogues. These are not supplements. They are drugs. The compounds that serious practitioners are running — sermorelin, BPC-157, TB-500, PT-141, CJC-1295, ipamorelin — sit somewhere on a regulatory and clinical spectrum that is neither “supplement” nor “approved pharmaceutical,” and that ambiguity is precisely what the last two years of legal reform have been grappling with.
If this framing is new to you, the first piece in this series covers the legal shift in more detail.
The Classes of Peptides Practitioners Actually Use

Not all peptides are used for the same purpose. The field has developed informal categories based on primary application, though most compounds have overlapping effects.
Growth Hormone Secretagogues
These peptides stimulate the pituitary gland to release growth hormone. They include GHRH analogues (sermorelin, CJC-1295, tesamorelin) and ghrelin mimetics also called GHRPs (ipamorelin, hexarelin, GHRP-2, GHRP-6).
The clinical rationale for their use comes from the established decline of growth hormone secretion with age — a phenomenon documented in longitudinal studies going back to the 1980s (Iranmanesh et al., Journal of Clinical Endocrinology & Metabolism, 1991). Tesamorelin is FDA-approved for HIV-associated lipodystrophy, which means it has a human RCT evidence base. The off-label use of other secretagogues for anti-aging and body composition lacks equivalent human trial data. That gap matters, and any serious practitioner should hold it clearly.
What secretagogues have going for them, mechanistically, is that they work through the body’s own feedback systems rather than bypassing them. Exogenous growth hormone suppresses endogenous production. Secretagogues stimulate endogenous production. The hypothalamic-pituitary feedback loop stays intact. Whether that translates to a meaningfully better safety profile in long-term human use is not proven — it’s a reasonable hypothesis.
Repair and Recovery Peptides
BPC-157 and TB-500 are the two most discussed compounds in this category.
BPC-157 (Body Protection Compound 157) is a synthetic peptide derived from a 15-amino-acid sequence found in human gastric juice. In rodent models, it has shown statistically significant effects on tendon-to-bone healing (Cerovecki et al., Journal of Orthopaedic Research, 2010), muscle tear repair, and gut mucosal integrity. There are no published human RCTs. The mechanism appears to involve upregulation of growth hormone receptor expression and modulation of the nitric oxide system.
TB-500 is a synthetic version of thymosin beta-4, a naturally occurring peptide involved in actin regulation, wound healing, and inflammation modulation. Animal studies have shown effects on cardiac tissue repair and wound closure (Goldstein et al., Annals of the New York Academy of Sciences, 2012). Human trial data is similarly sparse.
These are probably the most widely used compounds in the non-clinical practitioner community. I wrote specifically about a twelve-week BPC-157 protocol and what it did to a chronic shoulder injury here. The evidence picture for both compounds is predominantly preclinical. That doesn’t make them ineffective. It makes the human evidence thin.
Melanocortin Peptides
PT-141 (bremelanotide) is a melanocortin receptor agonist developed from melanotan II, originally synthesized as a tanning compound. It acts centrally — meaning in the brain — on MC3R and MC4R receptors involved in sexual arousal pathways. Unlike PDE5 inhibitors (Viagra, Cialis), it doesn’t work peripherally on blood flow. It modulates desire at the neurological level.
Bremelanotide was FDA-approved in 2019 for hypoactive sexual desire disorder in premenopausal women under the brand name Vyleesi. This is relevant: it has a human RCT evidence base (Kingsberg et al., Obstetrics & Gynecology, 2019). The off-label use of PT-141 — including compounded versions used by both men and women — draws on that same mechanism, though the approved application is narrower.
Metabolic Peptides
This category overlaps with mainstream medicine more than most practitioners realize. Semaglutide is a GLP-1 receptor agonist — a peptide drug. Tirzepatide is a dual GIP/GLP-1 agonist — also a peptide. Both are now among the highest-revenue drugs in pharmaceutical history. The explosion in compounded semaglutide over the past three years is the most visible instance of the pharmaceutical-to-compounding pipeline that defines much of the current regulatory landscape.
AOD-9604 is a peptide fragment (177-191) of human growth hormone that was investigated for fat metabolism without the IGF-1 elevation associated with full GH. Phase III human trials for obesity failed to demonstrate efficacy over placebo — development was halted after a 24-week trial of 536 subjects showed no significant weight loss versus placebo. This is an important data point: not every mechanistically interesting peptide translates to clinical outcomes.
What “Research Peptide” Actually Means — and What It Doesn’t
You will encounter the phrase “for research use only” on peptide supplier websites. You need to understand what this phrase does and doesn’t mean.
What it does: it provides a legal framing that positions the compound as a laboratory reagent rather than a drug or supplement, allowing sale without FDA drug approval or supplement regulation.
What it doesn’t do: it doesn’t say anything about the purity, sterility, or actual composition of the compound you receive. It doesn’t mean the compound has been validated for research use by any independent body. It doesn’t confer any safety assurance.
The research peptide market has historically had significant quality control problems. Independent testing has found products that are underdosed, overdosed, contaminated, or mislabeled. This is not speculative — testing services have published results showing substantial variance across vendors. The legal reforms covered elsewhere in this publication have begun addressing the compounding pharmacy pathway as an alternative, but the research peptide gray market continues to exist alongside it.
Understanding the difference between a compounded peptide dispensed by a 503A pharmacy under a valid prescription and a lyophilized powder purchased from a website is foundational knowledge for anyone entering this space. They are not the same thing. Treating them as equivalent is a significant error.
Why Any of This Matters Now
A friend who practices medicine told me once, years ago, that peptides weren’t like drugs — they were from the same family as insulin. I filed it away. Then I tore something in my wrist snowboarding, did the imaging, did the physio, and still couldn’t load it without pain. Alternative medicine is where injured people end up when conventional medicine runs out of answers. That’s how I came back to the word. And this time, when I read, the concept finally snapped: peptides don’t add new chemistry to the body. They activate chemistry the body already runs.
Peptides have existed as a research and clinical tool for decades. Insulin has been a peptide therapy since the 1920s. What changed in the last two years — as described in the first article in this founding sequence — is the regulatory architecture governing compounding access. For a meaningful window, a broader set of peptide compounds became legally accessible through compounding pharmacies for the first time in years, following policy shifts at the FDA.
That window may or may not persist. Regulatory posture on compounding is not stable. What the shift did was surface a question that the serious biomedical community has been sitting with for a while: there is a class of compounds with credible preclinical evidence, plausible human mechanisms, and essentially no human trial data because no pharmaceutical company has a financial incentive to fund those trials. The patent situation for many of these peptides makes large-scale drug development economically irrational.
This is a genuine problem in evidence-based medicine — not unique to peptides, but especially acute here. The practitioner community running these compounds is, functionally, generating the only longitudinal human data that exists for some of them. That’s a strange position to be in. It’s also not going to resolve itself quickly.
What you can do, as a practitioner entering this space, is hold the evidence hierarchy accurately. Know which compounds have human trial data (tesamorelin, bremelanotide, semaglutide). Know which have strong animal models and zero human trials (BPC-157, TB-500). Know which have human trial data that failed (AOD-9604). The molecules are not all in the same epistemic position, and collapsing them into a single “peptides work” or “peptides are unproven” frame misses everything important.
What Practitioners Are Actually Asking
What’s the difference between a peptide and a protein?
Size and convention, primarily. Peptides are typically defined as chains of fewer than 50 amino acids; proteins are longer. The distinction matters practically because smaller chains tend to have more specific receptor activity and are more amenable to synthesis and compounding. In clinical contexts, the term “peptide” usually refers to a short chain with a defined therapeutic target, while “protein” refers to a larger, more structurally complex molecule. The line isn’t sharp, but it’s useful.
Do peptides require injections?
Most therapeutically significant peptides are administered subcutaneously because they’re degraded by digestive enzymes before they can reach systemic circulation. There are exceptions — some peptides have been formulated for intranasal delivery (PT-141 has an intranasal form), and oral peptides are an active area of pharmaceutical research. For the majority of compounds discussed in this publication, subcutaneous injection is the standard delivery route.
Are peptides the same as steroids?
No. Steroids are a class of lipid-derived hormones — testosterone, estrogen, cortisol — that act primarily through intracellular receptors and affect gene transcription. Peptides are amino acid chains that act primarily through cell-surface receptors and signaling cascades. The physiological effects can overlap in some contexts (both can affect body composition, for instance), but the biochemistry, legal status, side effect profiles, and regulatory histories are completely different.
What does “subcutaneous injection” actually involve?
A subcutaneous injection goes into the fat layer just beneath the skin — not into muscle. The standard site is the abdomen, though the upper thigh and outer arm are also used. The needles used are short (typically 4-6mm) and thin (29-31 gauge), similar to what people with diabetes use for insulin. The injection itself is minimally painful for most people. The learning curve is procedural, not particularly physiological.
If a peptide is “naturally occurring,” does that make it safer?
No, and this framing should be treated with suspicion whenever you encounter it. BPC-157 is derived from a sequence found in human gastric protein — that doesn’t make synthetic exogenous administration equivalent to endogenous presence. Insulin is naturally occurring; an insulin overdose is fatal. The origin of a compound’s structure tells you something about its mechanism. It tells you nothing definitive about its safety profile at therapeutic doses in a clinical context.
The evidence base for most therapeutic peptides is honest about what it is: compelling preclinical data, sparse human data, and a practitioner community that is running real protocols with real bodies in a regulatory environment that is still finding its shape. That’s the actual picture. It is, depending on your tolerance for uncertainty, either a problem or an opportunity — probably both.